CLINICAL APPLICATION
1. Killing of Cancer and Transformed Cells in vitro
To test the effects of the MEBO regenerative nutrients on cancer cells, in vitro experiments were conducted in cultures containing typical cancer cell lines, including solid tumor cell lines, lymphoma and leukemia cell lines.
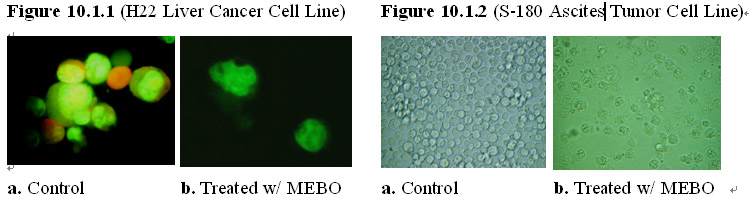
Typically, cancer cell lines were grown in normal culture medium and each cell line is divided into two groups: a treatment group and a control group. The MEBO regenerative nutrients were added to the cancer cell cultures in the treatment group while the cancer cell cultures in the control group were maintained without adding any supplemental materials. As shown in Figures 10.1.1-4, after 24 hours of exposure to the MEBO regenerative nutrients, solid tumor as well as leukemia cells in the treatment group manifested growth arrest with some cells showing the morphology of cell death. In comparison, the cancer cells in the control group proliferated at a normal rate and demonstrated normal cancer cell morphology. Prolonged exposure of the cancer cells (>48 hours) to the MEBO regenerative nutrients led to death of all cancer cells in the treatment group.

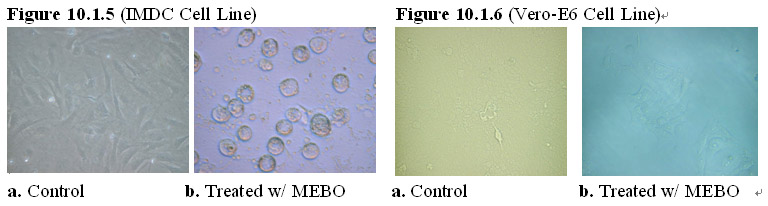
Interestingly, the MEBO regenerative nutrients also exert the same growth-arrest and cell-killing effects on transformed cells, such as an immortalized artery endothelial cell line (IMDC) and an immortalized kidney epithelial cell line (Vero-E6). As shown in Figures 10.1.5-6, after 24 hours of exposure to the MEBO regenerative nutrients, the transformed cells in the treatment group manifested growth arrest with some cells showing the morphology of cell death. In comparison, the transformed cells in the control group proliferated at a normal rate and demonstrated normal transformed cell morphology. Prolonged exposure of the transformed cells (>48 hours) to the MEBO regenerative nutrients led to death of all transformed cells in the treatment group.
2. Selective Killing of Cancer Cells Co-Cultured with Normal Cells
To test the effects of the MEBO regenerative nutrients on normal cells in the presence of cancer cells, in vitro co-culture experiments were conducted in a culture containing both normal bone marrow cells (small cells) and human gastric cancer cells (larger cells).
The co-culture was grown in normal culture medium (Figure 10.2.1a) and then divided into two groups: a treatment group and a control group. The MEBO regenerative nutrients were added to the co-culture in treatment group while the co-culture in the control group was maintained without adding any supplemental materials. As shown in Figure 10.2.1b, after 4 days of exposure to the MEBO regenerative nutrients, the cancer cells in the treatment group not only stopped growing but also manifested morphology of cell death. However, normal bone marrow cells in the treatment group not only survived but also proliferated vigorously. By day 7 virtually all cancer cells disappeared in the culture while the normal cells continued to proliferate (Figure 10.2.1c).
In comparison, in the absence of the MEBO nutrients by day 5 the cancer cells in the control group proliferated vigorously while the normal cells virtually disappeared in the culture (Figure 10.2.1d). By day 7, the cancer cells proliferated to reach confluency while no normal cells could be observed in the culture (Figure 10.2.1e).

3. Promoting Growth of Normal Cells in vitro
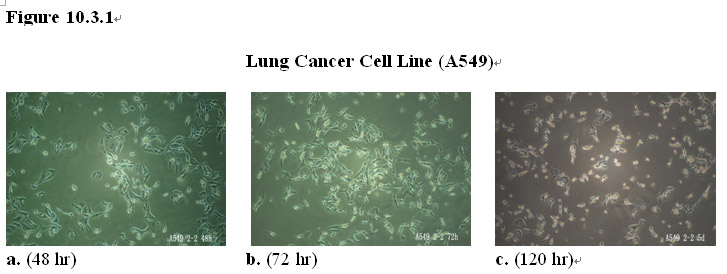
To further confirm the dual functions of the MEBO regenerative nutrients and selective cell-killing effects on human cancer cells, in vitro experiments were conducted in separate cultures containing A549 lung cancer cells (Figures 10.3.1a-c) and MRC5 normal lung fibroblasts (Figures 10.3.1d-f), respectively. The MEBO regenerative nutrients were added to both cultures.
After 48 hours of exposure to the MEBO regenerative nutrients, lung cancer cells manifested growth arrest and some cells showing morphology of cell death (Figure 10.3.1a). After prolonged exposure (>72 hours) virtually all cancer cells died in the culture (Figures 10.3.1b-c).
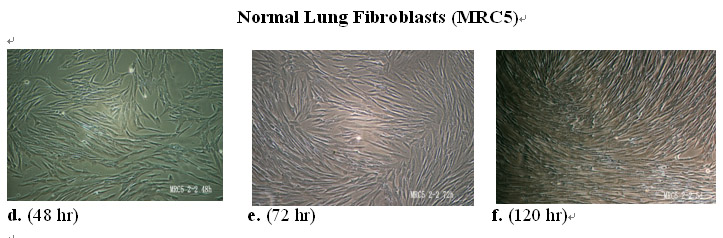
In contrast, in the presence of the MEBO regenerative nutrients normal lung fibroblasts proliferated vigorously and continued to do so to reach confluency by day 5 (Figures 10.3.1d-f).
4. Differential Metabolism of MEBO Regenerative Nutrients in Cancer and Normal Cells
To explore the mechanism of dual functions of the MEBO regenerative nutrients, examination of the cancer cells and normal cells under ultra-fine electron microscope was carried out. A549 lung cancer cells and MRC5 normal lung fibroblasts were cultured in the presence of the MEBO regenerative nutrients for 48 hours and then fixed for microscopic examination.
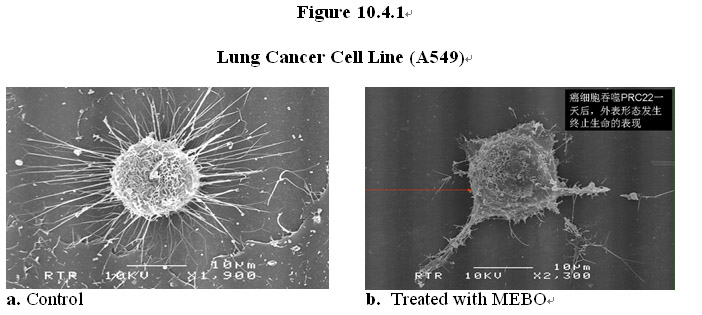
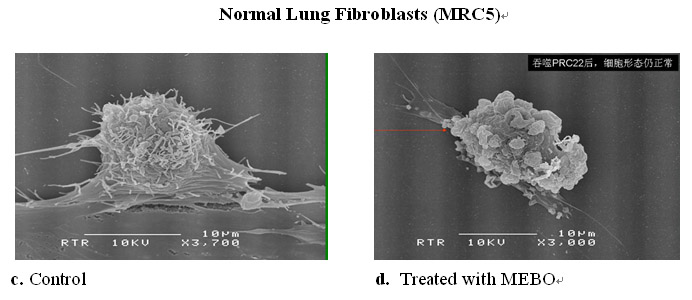
As shown in Figure 10.4.1 below, under a scanning electron microscope there is a dramatic difference in the cell surface morphology of the cancer before and after exposure to the MEBO regenerative nutrients. The rich microvilli extending from the surface of the cancer cell and attached to the culture plate (Figure 10.4.1a) virtually disappeared and detached (Figure 10.4.1b). In comparison, cell surface morphology in the normal cell appeared to be normal after the treatment with the MEBO regenerative nutrients (Figures 10.4.1c-d).
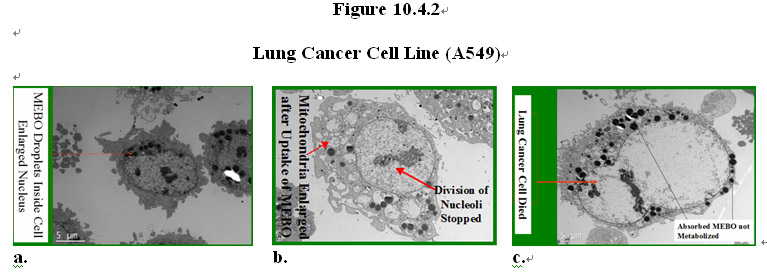
The intracellular components of the cancer and normal cells were examined under a transmission electron microscopy. Dramatic differences were also observed in the response of the cancer and normal cells to the MEBO regenerative nutrients. As shown in Figure 10.4.2, both types of cells readily absorbed the MEBO nutrients that are lipophilic, as shown by the dark dots of oil droplets inside the cells (Figure 10.4.2a). Surprisingly, in the cancer cell, upon uptake of the nutrients into the cytoplasm, it can be observed that mitochondria of the cancer cell were enlarged and swollen while the nucleus stopped dividing (Figure 10.4.2b). The nutrient droplets stayed in the cytoplasm and did not get metabolized before the growth arrest which was then followed by cell death (Figure 10.4.2c).
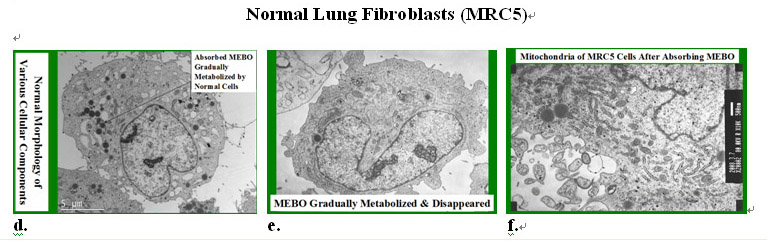
In contrast, in the normal cell the MEBO nutrient droplets were taken into the cytoplasm and gradually metabolized by the cell (Figures 10.4.2d-e). The normal cells not only did not die but also underwent vigorous proliferation as shown by active cell division and increase of cell numbers in the culture. Mitochondria of the normal cell appeared to be normal after uptake of the MEBO regenerative nutrients.
These results demonstrate distinctly differential metabolism of the MEBO regenerative nutrients in cancer and normal cells. The cancer cells absorb the nutrients, which causes enlargement and swelling of mitochondria, the power plants of a cell. As observed under the microscope, the cancer cells subsequently undergo growth arrest which is followed by cell death. It is conceivable that the substantial modification of the structure of mitochondria leads to disruption of their function, leading to a shutdown of the cellular activity and release of pro-apoptotic factors, causing cell death. Importantly, the growth of the normal cells is not negatively affected but instead promoted to more vigorous levels. Such dual functions of the MEBO regenerative nutrients could explain why the MEBO regenerative nutrients exhibit little toxicity yet possess efficacy in suppressing tumor growth in animal models and clinically.
Recently, it has been recognized that mitochondria play new critical roles in cell metabolism ― regulation of cell death pathways. Gogvadze et al. (2008) “Mitochondria in cancer cells: what is so special about them?” Trends in Cell Biol. 18:165-173. Wang (2008) “The expanding role of mitochondria in apoptosis” Genes & Dev. 15:2922-2933. It has been reported that several mitochondrial proteins that reside in the intermemebrane space of mitochondria, in response to a variety of apoptotic stimuli, are released to the cytosol and/or the nucleus, thereby directly activating cellular apoptotic program and causing apoptosis of the cell. Liu et al. (1996) “ Induction of apoptosis program in cell-free extracts: requirement for dATP and cytochrome c” Cell 86:147-157. Verhagen et al. “Identification of DIABLO, a mammalian protein that promotes apoptosis by binding to and antagonizing IAP proteins” Cell 102:43-53. Based on our EM data obtained, the cancer cell death induced by the MEBO nutrients can be attributed to activation through disruption of mitochondrial membrane and subsequent releasing of the apoptotic proteins. In contrast, normal cells in the presence of the MEBO nutrients not only survive but also grow rigorously.
A number of agents, including chemotherapeutic drugs (e.g., cisplatin and arsenic oxide) and compounds isolated from natural sources (e.g., epigallocatechin gallate (EGCG) and beta-carotene) are shown to be able directly or indirectly target mitochondria, the “power plants” of the cell. Pederson (2007) “The cancer cell’s “power plants” as promising therapeutic targets: An overview” J. Bioenerg. Biomembr. 39: 1-12. However, there has been no report of these agents having a dual function of killing cancer cells while promoting normal cell growth and regeneration, as demonstrated by the MEBO nutrients. It is well known that virtually all of anti-cancer chemotherapeutic agents can cause the side effects of suppressing the growth of normal, fast dividing cells, such as epithelial cells in the skin, hair follicles and mucosa, as well as that of the hematological cells.
It is plausible that the intrinsic differences in the metabolism of normal and cancer cells may contribute to the differential response to the MEBO nutrients. It has been observed that cancer cells produce most of their ATP through glycolysis, even under aerobic conditions, and there is a correlation between glycolytic ATP production and aggressiveness of the tumor cells. Warburg (1930) “The metabolism of tumors” London: Arnold Constable. Simonnet et al. (2002) “Low mitochondrial respiratory chain content correlates with tumor aggressiveness in renal cell carcinoma” Carcinogenesis 23:759-768. When rapidly growing tumors shift their ATP production to glycolysis, mitochondrial activity slows down. Gogvadze et al. (2008), supra. By absorbing the highly lipophilic MEBO nutrients, the cancer cells seem to be unable to metabolize them; instead, the undigested lipid droplets stayed inside the cytoplasm, which caused swelling of mitochondria, leading to cell death (Figure 10.4.2c). In contrast, in the normal cell the nutrient droplets were taken into the cytoplasm and gradually metabolized by the cell (Figures 10.4.2d-e); mitochondria in the normal cells did not appear to be damaged by the uptake of the nutrients and continued to function normally, if not more vigorously. As demonstrated in the skin and mucosal tissue regeneration of wound and ulcer patients, the MEBO nutrients consistently promote physiological repair and regeneration of normal cells in humans, including fully developed adult humans.
The unique ability of the MEBO nutrients to promote normal cell growth without damaging the cellular power plants―mitochondria (as demonstrated in their clinical efficacy in promoting tissue and organ regeneration and in in vitro cellular studies) and their surprising ability to induce apoptosis of transformed and cancer cells are extremely desirable in the development of safe and efficacious products for cancer treatment and prevention. As MEBO nutrients are formulated using natural botanicals safely used for human consumption for thousands of years, these products have superior safety profiles and can be safely used a nutritional support for cancer patients, which forms the scientific foundation for MEBO’s ongoing large population human studies of end-stage cancer patients. See Section 10.10 below. This innovative approach to cancer treatment and prevention will open a new fertile field for basic and clinical applications.
5. Killing of Cancer Cells in Tumor Tissue in vitro
To test the effects of the MEBO regenerative nutrients on cancer cells in solid tumors, in vitro experiments were conducted in a culture containing explants from human solid tumor tissues.
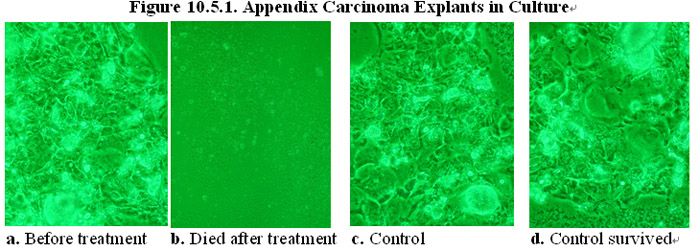
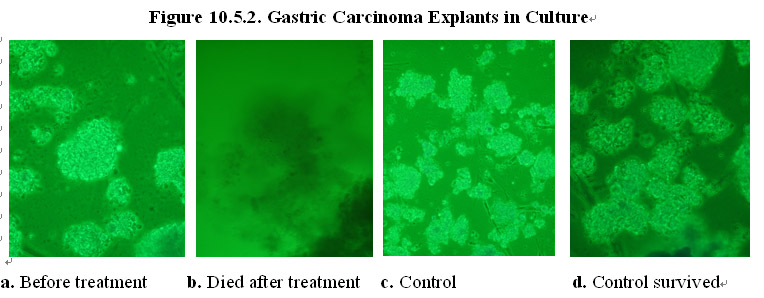
As shown in Figures 10.5.1a and c, explants of human appendix carcinoma tissues were divided into treatment and control group and cultured in normal cell culture medium. In the presence of the MEBO regenerative nutrients, the cancer cells in the treatment group died and the tumor tissue explants disintegrated in the culture (Figure 10.5.1b). In comparison, in the absence of the MEBO regenerative nutrients, the cancer cells in the control group did not die and the explants survived in the culture (Figure 10.5.1d).
In another experiment, explants of human gastric carcinoma tissues were divided into treatment and control group and cultured in normal cell culture medium (Figures 10.5.2a and c). In the presence of the MEBO regenerative nutrients, the cancer cells in the treatment group died and the tumor tissue explants disintegrated in the culture (Figure 10.5.2b). In comparison, in the absence of the MEBO regenerative nutrients, the cancer cells in the control group did not die and the explants survived in the culture (Figure 10.5.2d).
6. Suppression of Ascites Tumor Growth in Animal Tumor Models
To test the effects of the MEBO regenerative nutrients on the growth of cancer cells in vivo, a standard animal model for highly aggressive malignant cancer growth, mice bearing S180 ascites tumors, was used. When injected into the peritoneal cavity, S180 cells grow exponentially with a generation time of 12-18 hours. Growing primarily in suspension in the peritoneal cavity, S180 cells gradually plug peritoneal lymphatic drainage, leading to accumulation of ascites fluid within 2 weeks. S180 cells can also metastasize into major organs near the peritoneal cavity, such as the liver, kidney, pancreas, lung, stomach, and intestine. Mice that develop ascites normally die in 3-4 weeks. S180-induced ascites represents one of the most aggressive transplantable cancers in experimental mouse models.
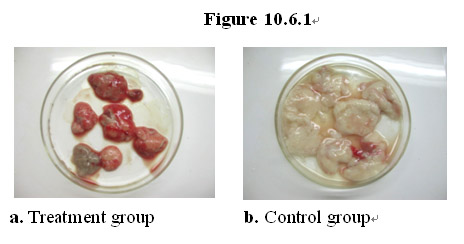
In the study, fourteen (14) mice were injected intraperitoneally with S180 cells to induce formation of ascites tumors and 15 days later divided equally into treatment and control group. In the treatment group, seven (7) rats were fed with a regular diet supplemented with the MEBO regenerative nutrients while the control group with just the regular diet. Seventeen (17) days later the mice from both groups were sacrificed and ascites tumors were removed from the peritoneal cavity and weighed.
As shown in Figure 10.6.1a, the average weight from the tumors from the treatment group is about 1 g. In contrast, the average weight from the tumors from the control group is about 3 g (Figure 10.6.1b). These results demonstrate that the MEBO regenerative nutrients are active in the suppression of aggressive tumor growth in vivo.
7. Eradication of Skin Cancer in Patients
Topical formulations of the MEBO regenerative nutrients were used to successfully treat various forms of skin cancer, including basal cell carcinoma and squamous cell carcinoma.
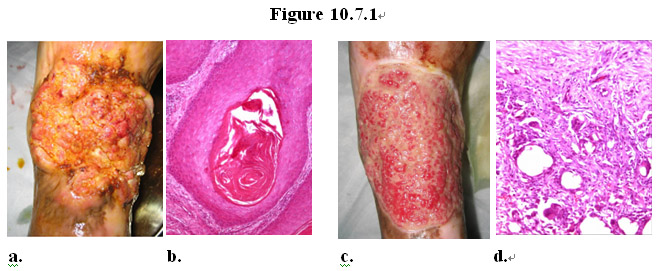
As shown in Figure 10.7.1, a patient with squamous cell carcinoma (Figure 10.7.1a) on his leg was treated with a topical formulation of the MEBO regenerative nutrients. Twenty (20) days later pathological examination of the tissue at the cancer site revealed that the original cancerous tissue (Figure 10.7.1b) disappeared, replaced with normal healthy granulation tissue (Figure 10.7.1 c-d).
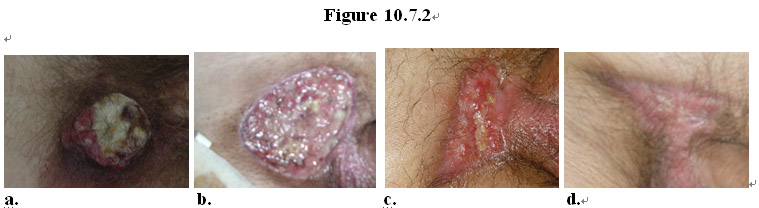
As shown in Figure 10.7.2, a patient with low differentiated squamous cell carcinoma (Figure 10.7.2a) near his genital was treated with a topical formulation of the MEBO regenerative nutrients. Ten (10) days later pathological examination of the tissue at the cancer site revealed that the original cancerous tissue disappeared, replaced with normal healthy granulation tissue (Figure 10.7.1b-c). Continued treatment with the MEBO product resulted in eradication of the cancerous tissue and healing of the wound (Figure 10.7.1d).
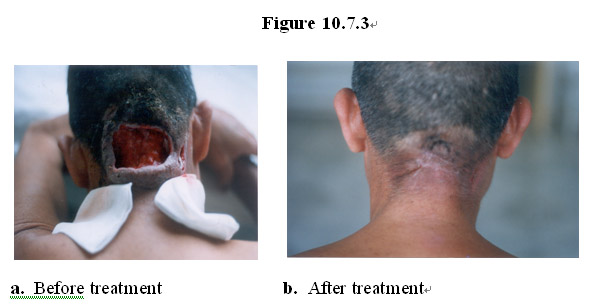
As shown in Figure 10.7.3, a patient with severe erosive, necrotic wound caused by basal cell carcinoma (Figure 10.7.3a) on his neck was successfully treated with a topical formulation of the MEBO regenerative nutrients, resulting in complete eradication of cancerous tissue and healing of the gapping open wound (Figure 10.7.3b).
8. Eradication of Leukemia in Patients
Oral formulations of the MEBO regenerative nutrients have been used as nutritional support for end-stage cancer patients who have given up medical treatment of cancer.
As shown in Figure 10.8.1, a patient (female, age 43) was diagnosed with acute myeloid leukemia (AML-M2a) 6 years ago and went into remission after treatment with chemotherapy. In October 2007 her leukemia reoccurred and was resistance to treatment with chemotherapy. As shown in Figure 10.8.1a, peripheral blood and bone marrow smear tests showed that cells in the granulocyte series accounted for 35.5% with the myeloblast count in her bone marrow increased to 25.5% with varied sizes and irregular morphology.
Starting January 2008 she gave up medical cancer treatment and started taking the MEBO regenerative nutrients as nutritional support. One and a half months later, peripheral blood and bone marrow smear tests showed that cells in the granulocyte series accounted for 44% with no myeloblast detected, and cells in all phases showed generally normal ratios and morphology; red blood cell series accounted for 26%; and cells in all phases showed generally normal ratios and morphology (Figure 10.8.1b). After 3 months on the MEBO regenerative nutrients, her overall condition improved significantly with healthier complexion, better quality sleep, and gained about 5 Kg of weight (Figure 10.8.1c).

These results demonstrate that systemic uptake of the MEBO regenerative nutrients can cause complete remission of relapsed AML and full restoration of normal hematological cell counts, ratios and morphology. Such results are extremely encouraging and shed new light into innovative treatment of late-stage leukemia without intervention using chemotherapy or hematological/stem cell transplantation. The fact that the patient’s hematological cell counts, ratios and morphology have been fully restored suggests that the MEBO regenerative nutrients may act on the source of all hematological cells: the hematopoietic stem cells, by promoting proliferation and differentiation of the stem cells that follow the correct, normal lineages while inducing death of leukemia cells, thereby regenerating the hematological system and eradicating cancer. Further human studies led by MEBO are under way in larger patient populations in China.
9. Regeneration of internal organs of cancer patients
Oral formulations of the MEBO regenerative nutrients have also been used to restore structure and function of tissues and organs of cancer patients that are damaged by the side effects of chemotherapy and radiation therapy.
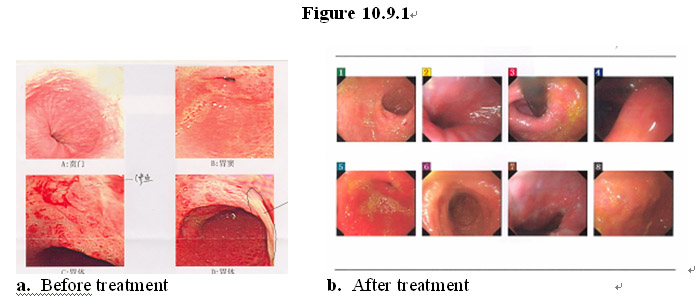
As shown in the gastrointestinal endoscopic images in Figure 10.9.1a, a patient (male, age 59) had pancreatic cancer that had metastasized to the stomach and sustained extensive damage of the gastric lining due to chemotherapy and radiation, leading to the development of erosive gastric ulcers. After taking the MEBO regenerative nutrients, his gastrointestinal wall and other GI lining have been regenerated with normal, healthy mucosa and underlying tissues (Figure 10.9.1b).
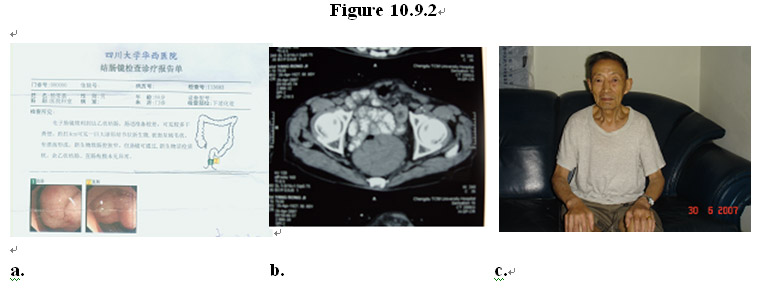
In another example, as shown in the gastrointestinal endoscopic images in Figure 10.9.2a-b, a patient (male, age 80) was diagnosed with rectal cancer with the tumor mass located at 4 cm above the anus, causing ulcer lesion there. After taking the MEBO regenerative nutrients for 3 months the tumor disappeared and the rectum is regenerated with normal, healthy mucosa and underlying tissues (Figure 10.9.2c).
10. Ongoing Clinical Studies of End-Stage Cancer Patients
Formally commenced on Janurary 25, 2008 in China, MEBO initiated a large-scale clinical studies of end-stage cancer patients who had failed all medical interventions and given up upon further medical treatment (as certified by patients’ oncologists). About 300 end-stage cancer patients, exhibiting a variety of different cancer types (including lung, liver, gastrointestinal (esophagus, stomach, colon, rectal cancer), pancreas, breast, prostate, uterus/cervix, ovarian, thymus, neuroblastoma) were enrolled and provided with MEBO regenerative nutrients. Preliminary results are extremely encouraging: most patients (~80% overall 1-year survival rate) not only survived but also improved significantly in all detectable indexes including the body weight, reduction in tumor size, elimination of leukemia cells (in patients with relapsed leukemia), and improvement in mobility and vitality.
For example, a 58 year-old female patient was diagnosed with right lung cancer that had metastasized to the bone, and was treated with chemotherapy for 3 courses without surgical operation. The treatment modality did not improve her condition and she still coughed up blood (haemoptysis) and had a persistent pain in her right ribs. She subsequently gave up upon such treatment and started taking the MEBO regenerative nutrients in February 2008. Four months after her overall condition improved: haemoptysis disappeared; rib pains alleviated; and appetite, complexion and vitality improved (Figure 10.10.1a). The tumor in her right lung regressed from a size of 30x43 mm to 27x29 mm.
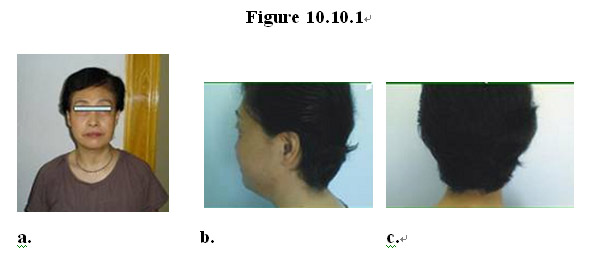
Remarkably and surprisingly, after 4 months on the MEBO regenerative nutrients the patient who had suffered from hair loss due to chemotherapy started grow thick, lush and lustrous black hair (Figure 10.10.1b-c) that appeared to be even healthier than a typical woman of her age. The texture of her skin also improved, becoming suppler and smoother. Interestingly, her breasts seemed to become firmer and vaginal discharge that had disappeared 7-8 years ago reappeared.
Currently these patients have voluntarily elected to continue getting nutritional support from MEBO and their monthly reports are gathered by MEBO for further analysis.
The results obtained from MEBO’s 1-year clinical studies, although preliminary, are very encouraging. They demonstrate that by using the MEBO regenerative nutrients as nutritional support, cancer patients, even if at a late or end stage, can still benefit from this unique modality in the absence of conventional medical intervention or after failure of all medical interventions. The patients not only survived but also enjoy better quality of life. In some cases, such as relapsed acute leukemia and gastrointestinal cancer, the patients have had complete remission and remarkably improved vital indexes. Continuous taking of the MEBO nutrients also seemed to have certain rejuvenating effects on the patients who manifested certain degree of tissue/organ regeneration during the course, such as growing healthier-, younger-looking skin and hair.
These preliminary results obtained by MEBO in its cellular, tissue and human studies should shed new light on cancer research, prevention and treatment. Since the 1950’s, despite the enormous amounts of funding targeted for cancer research by the governments and private entities throughout the world, it is yet far away from claiming victory in the ongoing war against cancer. It has been predicted that one in two men and one in three women are likely to die of cancer. World Fact Book, Central Intelligence Agency, Office of Public Affairs, Washington, D.C., July 2006. Thus, it can be estimated that, if anti-cancer agents with long-term curative effects are not found soon, particularly for advanced metastatic cancers, about 2.7 billion people would die of cancer, considering there are about 6.5 billion people in the world today. In the United States, and in the rest of the world, people are still looking for the first “Magic Bullet” that will consistently destroy in different patients both primary and metastatic cancers regardless of their tissue of origin while exhibiting minimal toxicity to the human host. Pederson (2007), supra.
Against this grim background, MEBO beckons as a bright beacon for the world. Built upon solid scientific and clinical foundations and with superior safety profiles, the MEBO approach of utilizing its dual-functional regenerative nutrients and technologies offers enormous hope for humanity as a whole, and globally it will have a profound impact on the currently overburdened health care systems and overstressed economies in both developed and developing countries.

